I’ve received a *lot* of questions over the past few days after several major news outlets posted surprising news: The cause of PCOS has finally been discovered, and it’s high AMH (Anti-Müllerian hormone) during pregnancy.(1)
This fascinates me because in addition to being a total nerd about PCOS, years ago,I wrote about the increased levels of AMH in women with PCOS.and it’s a hormone level I consistently test in my patients.
To summarize, the study found that women with PCOS have higher levels of AMH in pregnancy, and administering AMH injections to pregnant mice could induce PCOS in their offspring.
They also found that administering a commonly used IVF drug (cetrotelex) to the animals during pregnancy, as well as later in life, could to reverse these effects.
When these news outlets asserted that finally, the cause of (and cure for) PCOS has been found, it caused many women to celebrate – it is definitely exciting to read a headline like this!
As is often the case, after reading the study in its entirety, it’s clear that what the media reported is not actually what the researchers concluded.
The findings shared in study are very exciting overall, but before we get led down the rabbit hole of headlines, let’s take a look at the facts.
What We Know About Prenatal Hormone Exposure and PCOS:
It is already well-established that giving testosterone to pregnant animals causes PCOS in their offspring.(2)
We are also aware that exposing fetuses to testosterone in the womb can permanently impact the homone-controlling GnRH neurons in their brains, causing irregular ovulation when the animals reach puberty.(3)
And it’s not just about exposing unborn babies to actual hormones like testosterone – it’s also been found that a single exposure to bisphenol A (found in plastics) and PCE (an industrial pollutant) can induce PCOS in the offspring for generations(4).
These chemicals have endocrine disruption effects with clear estrogen-like actions on the developing baby’s brain and reproductive organs.
Overall one thing is clear: we are accumulating a LOT of evidence showing that the hormonal environment during pregnancy has long-term and often permanent effects on the hormonal systems of babies, and it seems that AMH is no different.
AMH in Pregnancy in PCOS: Lean Vs. Classic
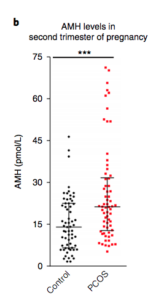
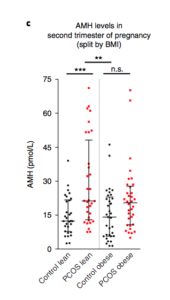
Researchers in this study found that pregnant women with lean PCOS had a statistically higher AMH level than lean women without the condition.
This unfortunately, wasn’t found to be the case for women with PCOS of higher body mass index (Classic PCOS), their AMH in pregnancy was more similar to that of women without PCOS
However, to be fair, even in classic PCOS -you’ll see in the figure below that there *was* a slight increase in AMH in pregnancy compared to those without the condition, even though it didn’t reach statistical significance in the study.
What the Study Discovered About Rodent Models and AMH in Pregnancy:
As mentioned, in this study researchers injected healthy pregnant rodents with AMH, which ended up causing PCOS-like traits such as delayed ovulation in their offspring.
The offspring mice did NOT, however, develop any of the metabolic abnormalities, weight gain, or insulin resistance that are characteristic of PCOS.
These metabolic characteristics are found in the majority of women with PCOS – both lean and classic to varying degrees.
Levels in Human Pregnancy Compared to Those in Study
Let’s look first at the dosing of AMH (0.12mg/100g/D) used on the mice in this study to see if this matches up with the increased amount found in women with PCOS in pregnancy.The researchers did not measure the serum levels of these animals, and they didn’t provide any reasoning for choosing the dose they did.
First, from other research (of which there is not very much), we know that injecting mice with double the amount of AMH used in this study causes a 16 fold rise in the animals’ blood levels. So, we know that it is most likely that injecting mice with around half the amount could potentially cause an 8-fold rise in their levels.
Of course, this won’t be exactly accurate but we would expect it to be somewhat in the same ballpark.

Regardless, even if this approximation is not quite accurate, we can clearly see that there is nowhere near an 8-fold difference in the majority of women with PCOS’ AMH levels in pregnancy compared to control women (see below).
On average, women with PCOS scored around 5-6 pmol/L higher in pregnancy compared to controls, just around a 1.3 fold increase compared to control women.
In women with a higher body mass index who have Classic PCOS, the difference is less between them and the control group.
So overall, the mice were injected with an amount of AMH that would raise levels much higher than we would see in PCOS in pregnancy.
So this is not necessarily a model for how PCOS is caused in pregnant women, but rather another model for how the injection of a very high amount of AMH can create a PCOS-like hormonal disruption in babies.
In fact, the function of AMH as a hormone is to slow and suppress the development of follicles in the ovary. It makes complete sense that injecting a pregnant animal with a massive dose would interrupt ovulatory function in the offspring in a similar way to that of other hormonal disruptions in pregnancy.
Insulin Resistance and Androgen Excess Links in Pregnancy
We are well aware that high levels of testosterone in pregnancy can induce PCOS in children, including BOTH the metabolic and androgen excess characteristics.(5)
We also know that high testosterone in pregnancy cause gut microbiota changes in the offspring that are characteristic of the metabolic abnormalities found in PCOS.(6)
Research also tells us that working on insulin resistance in PCOS pregnancy can prevent the GnRH abnormalities found in animal models of PCOS, and lower testosterone levels.(7).
Overall, it’s clear that prenatal metabolic influences as well as reproductive hormones have a role to play in the development of PCOS.
Other Sources of Prenatal Androgens in PCOS
It has also recently been discovered that 11-OH adrenal androgens are the most abundant androgens in women with PCOS, andare not affected by AMH from what we know currently. There have been no studies of these hormones in pregnant women with PCOS, so it is difficult to know what role they might play.
It is also well-known that elevated androgens *themselves* increase the production of AMH by the the ovary. Excess androgens cause the granulosa cells of the ovary to over-express AMH in response to FSH.(8)
As a result, it is difficult to tell what is cause and what is effect in a woman with PCOS who is pregnant.
Insulin Resistance Can’t Be Ignored in a Cure for PCOS
Finally, and possibly most importantly this study doesn’t explain ANY of the metabolic effects so strongly linked to PCOS, as giving a massive injection of AMH to the pregnant mice still did NOT induce any of the metabolic abnormalities or insulin resistance that are so characteristic of the vast majority of women with the condition.
It would be impossible to state we have finally found the cause of and cure for this condition without covering these areas since they affect more than 85% of women with PCOS!
It is well known that women with PCOS can begin spontaneously ovulating as they get older due to reduced androgens – yet they continue to secrete more insulin and sustain the risks of cardiovascular disease, insulin resistance, type-two diabetes though their entire lifespan — even well after menopause.
Genetics Updates: Mother and Father
These media reports also imply that only the maternal environment plays a role in the development of PCOS.
That said, a great deal of research suggests a link to PCOS through fathers as well. We know that elevations in androgens are found in the cord blood of both male and female offspring born to mothers with PCOS(9). This means that males could easily be affected by androgen excess in utero.
Several studies also associate PCOS with male pattern baldness in male relatives.(10)
Interestingly as well, we know that a father’s obesity can induce metabolic disturbances in two generations of mice offspring!(11)
And a strong correlation has been found between fathers who have metabolic issues, and daughters who have PCOS.(12)
Since numerous studies have found paternal links to PCOS, this would have to be transmitted through genetic or epigenetic influences to children since the father doesn’t determine the in-utero environment .
For a good summary of the genetics of PCOS please seehere.
We Still Have a Lot To Learn
Since fetal programming in PCOS is affected by so many factors ranging from androgens, AMH, insulin resistance, genetics and environmental endocrine disruptors, we still have much to learn and more research must be done.
PCOS can’t bepermanently solved with a simple course of an IVF medication — the impacts of PCOS do not end when women have children or go though menopause.
Even women with PCOS who ovulate regularly suffer with not only the metabolic effects of insulin resistance and inflammation, but also androgen excess.
Conclusions and Takeaways
This study is groundbreaking and interesting, unraveling important pieces of the puzzle as we work to understand the multifactorial nature of PCOS.
As amazing as this study is, a few takeaways from all of this are certain: it’s important to increase the public’s awareness that PCOS is much more than anovulation and infertility.
Finally, anything we can do to help women manage PCOS both before conception and during pregnancy can have the biggest impacts not only on our own health, but especially for that of our future generations!
Article References:
- Tata, B., Mimouni, N. E. H., Barbotin, A.-L., Malone, S. A., Loyens, A., Pigny, P., … Giacobini, P.Elevated prenatal anti-Müllerian hormone reprograms the fetus and induces polycystic ovary syndrome in adulthood.Nature Medicine2018;
- Xu, N., Kwon, S., Abbott, D. H., Geller, D. H., Dumesic, D. A., Azziz, R., … Goodarzi, M. O.Epigenetic Mechanism Underlying the Development of Polycystic Ovary Syndrome (PCOS)-Like Phenotypes in Prenatally Androgenized Rhesus Monkeys.PLoS ONE2011;6(11):e27286.
- Berg, T., Silveira, M. A., Moenter, S. M.Prepubertal Development of GABAergic Transmission to Gonadotropin-Releasing Hormone (GnRH) Neurons and Postsynaptic Response Are Altered by Prenatal Androgenization.The Journal of Neuroscience2018;38(9):2283-2293.
- Nilsson, E., Larsen, G., Manikkam, M., Guerrero-Bosagna, C., Savenkova, M. I., Skinner, M. K.Environmentally induced epigenetic transgenerational inheritance of ovarian disease.PloS One2012;7(5):e36129.
- de Melo, A. S., Dias, S. V., Cavalli, R. d. C., Cardoso, V. C., Bettiol, H., Barbieri, M. A., … Vieira, C. S.Pathogenesis of polycystic ovary syndrome: multifactorial assessment from the foetal stage to menopause.Reproduction2015;150(1):R11-R24.
- Sherman, S., Sarsour, N., Salehi, M., Schroering, A., Mell, B., Joe, B., Hill, J. W.Prenatal Androgen Exposure Causes Hypertension and Gut Microbiota Dysbiosis.Gut Microbes2018;01-45.
- Cardoso, R. C., Burns, A., Moeller, J., Skinner, D. C., Padmanabhan, V.Developmental Programming: Insulin Sensitizer Prevents the GnRH-Stimulated LH Hypersecretion in a Sheep Model of PCOS.Endocrinology2016;157(12):4641-4653.
- Dewailly, D., Robin, G., Peigne, M., Decanter, C., Pigny, P., Catteau-Jonard, S.Interactions between androgens, FSH, anti-Müllerian hormone and estradiol during folliculogenesis in the human normal and polycystic ovary.Human Reproduction Update2016;22(6):709-724.
- Daan NM, Koster MP, Steegers-Theunissen RP, Eijkemans MJ, Fauser BC.Endocrine and cardiometabolic cord blood characteristics of offspring born to mothers with and without polycystic ovary syndrome.Fertil Steril2017 Jan;107(1):https://www.omim.org/entry/184700#5.
- Fullston T, Ohlsson Teague EM, Palmer NO, DeBlasio MJ, Mitchell M, Corbett M, Print CG, Owens JA, Lane M.Paternal obesity initiates metabolic disturbances in two generations of mice with incomplete penetrance to the F2 generation and alters the transcriptional profile of testis and sperm microRNA content.FASEB J2013 Oct;27(10):4226-43.
- Leibel, N. I., Baumann, E. E., Kocherginsky, M., Rosenfield, R. L.Relationship of Adolescent Polycystic Ovary Syndrome to Parental Metabolic Syndrome.The Journal of Clinical Endocrinology Metabolism2006;91(4):1275-1283.