The Pregnenolone Steal
A Closer Look at this Popular Concept
By Dr. Fiona McCulloch, ND: author of 8 Steps to Reverse Your PCOS and peer reviewer of the 2023 International Evidence-based PCOS Guidelines
Published March 2020 | Updated March 2026
What is the Pregnenolone Steal?
The pregnenolone steal has been taught in the world of functional and naturopathic medicine for many years. Although it seems to provide a tidy explanation of how stress can impact sex hormones, it is important to re-examine its validity at a time when our understanding of steroidogenesis has advanced considerably.
Pregnenolone is synthesised from cholesterol and sits at the top of the steroid hormone cascade. From pregnenolone, the body can produce progesterone, DHEA, cortisol, estrogen, and testosterone, which is why it is often called the “mother hormone.” The pregnenolone steal theory proposes that during times of stress, the body diverts pregnenolone preferentially toward cortisol production. Because cortisol and progesterone (and other sex hormones) share this common precursor, the theory holds that making more cortisol necessarily leaves less pregnenolone available for sex hormones. The result, according to the theory, is that stress literally steals your hormonal raw material, leaving you progesterone-deficient and fatigued.
It is an appealing idea. Stress is real. Hormonal symptoms are real. Connecting the two through a single upstream mechanism makes intuitive sense, and for patients trying to make sense of their fatigue, low libido, irregular cycles, or insomnia, it offers a coherent story. But coherent stories and accurate biochemistry are not always the same thing.
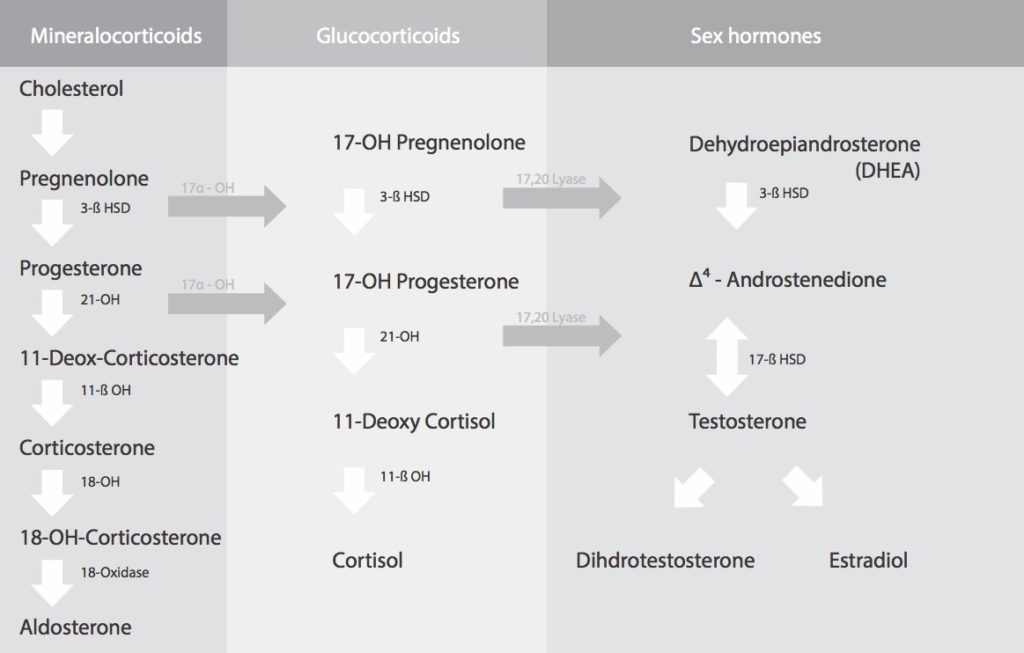
The full steroidogenesis cascade. Cholesterol converts to pregnenolone, which then branches into three pathways: mineralocorticoids (including aldosterone), glucocorticoids (including cortisol), and sex hormones (including testosterone and estradiol). The pregnenolone steal theory assumes these pathways compete for a shared pool — the diagram above shows they share a common origin, which is where the theory’s appeal comes from.
Let's Look a Little Deeper
The Compartmentalization Problem
The pregnenolone steal theory rests on a foundational assumption: that there is a single, shared pool of pregnenolone that the body draws from to produce both cortisol and sex hormones. When demand for cortisol rises, the theory goes, that shared pool gets diverted. This assumption is the central flaw.
Steroidogenesis does not work from a shared reservoir. Pregnenolone is synthesised locally — within individual steroidogenic tissues — from cholesterol, using the enzyme CYP11A1 (cholesterol side-chain cleavage enzyme) located in the inner mitochondrial membrane. This process occurs independently in the adrenal cortex, the ovarian theca and granulosa cells, and to a lesser extent in the brain and placenta. Each tissue produces its own pregnenolone under its own regulatory signals.
The adrenal cortex produces pregnenolone in response to ACTH (adrenocorticotropic hormone), released by the pituitary when the body is under stress. The ovaries produce pregnenolone in response to LH and FSH — the gonadotropins that govern the menstrual cycle. These are entirely separate regulatory systems, operating in separate tissues, responding to separate hormonal signals. There is no pipeline connecting adrenal pregnenolone production to ovarian sex hormone production. The adrenals cannot “steal” from the ovaries because they were never drawing from the same source.
The true rate-limiting step in steroidogenesis is not pregnenolone availability — it is the transport of cholesterol into the mitochondria by the steroidogenic acute regulatory protein (StAR). StAR expression is tissue-specific and independently regulated in each steroidogenic cell type. When the adrenals receive an ACTH signal to make more cortisol, they upregulate StAR and CYP11A1 expression locally. This increase in adrenal steroidogenesis does not deplete ovarian pregnenolone because the ovaries were never contributing to adrenal pregnenolone production in the first place.

Adrenal and ovarian steroidogenesis are separate, independently regulated processes. Each tissue converts cholesterol to pregnenolone under its own signals — ACTH drives adrenal production, while LH and FSH drive ovarian production. There is no shared pregnenolone reservoir between them. Diagram: Dr. Fiona McCulloch, ND.
The Real Mechanism: How Stress Actually Affects Your Hormones
So if the pregnenolone steal is biochemically inaccurate, how does stress affect reproductive hormones? The answer lies not in the adrenal gland but in the hypothalamus — the region of the brain that orchestrates the entire hormonal cascade.
Chronic or significant stress activates the hypothalamic-pituitary-adrenal (HPA) axis, triggering the release of corticotropin-releasing hormone (CRH) from the hypothalamus. CRH has a direct inhibitory effect on gonadotropin-releasing hormone (GnRH) pulsatility — the rhythmic signal that drives the entire reproductive hormone axis. When GnRH pulses are suppressed, the pituitary releases less LH and FSH. Without adequate LH and FSH signalling, follicle development slows, ovulation may not occur, and the corpus luteum — the structure that produces the majority of a woman’s progesterone — either fails to form or produces less progesterone than it would under optimal conditions.
This is called functional hypothalamic amenorrhea (FHA), and it is the primary mechanism by which chronic stress suppresses reproductive hormones. Crucially, it is a centralised neurological response originating in the brain, not a peripheral substrate competition happening in the adrenal gland.
There is also an important quantitative point that often gets overlooked in discussions of the pregnenolone steal. The amount of progesterone produced by a normal ovulatory cycle — secreted by the corpus luteum after ovulation — is substantially greater than any amount that could be diverted from a shared pregnenolone pool, even if such a pool existed. The scale of progesterone production from ovulation dwarfs what the steal mechanism could theoretically account for. This is one reason why, in clinical practice, the solution to low progesterone is typically either to support ovulation or to replace progesterone directly — not to address a substrate competition.
Stress can also affect the ovary more directly through elevated cortisol, which impairs the sensitivity of ovarian cells to LH and FSH signalling. But again, this is a direct hormonal effect on receptor sensitivity and cellular response — not a competition for pregnenolone substrate.
The cortisol-progesterone relationship, in other words, is real. But the mechanism is brain-based and deliberate, not substrate-based and passive. As I explain to patients in clinical practice: reproduction takes a significant amount of energy. The human brain is naturally designed to reduce investment in reproduction during periods of extreme stress or food scarcity. This is an adaptive, intentional response: the brain conserving energy by temporarily deprioritising one of the most metabolically costly functions it oversees. It is not your hormones stealing from each other. It is your brain making a calculated trade-off.
So What IS Happening to Your Hormones?
If you came to this article because you’ve been told — or suspected — that the pregnenolone steal is behind your fatigue, low progesterone, insomnia, or heavy periods, I want to address something directly: your underlying concern is well-founded. Something real is changing with your hormones. The pregnenolone steal oversimplifies the mechanism, but the clinical observations it attempts to explain are genuine. The question worth asking is not “is stress affecting my hormones?” (the answer to that is yes, it can). The better question is: what is actually driving those changes in your specific situation, and what can be done about it?
In my clinical practice, the mechanisms I most commonly see behind low progesterone, altered cycles, and related symptoms fall into three distinct categories — each with a different clinical picture and a different approach:
- Perimenopause: Progesterone decline is driven by the natural aging of the ovaries — specifically, by changes in egg quality and ovarian reserve that reduce how often ovulation occurs. This is the most common mechanism I see in patients in their 40s, and it has nothing to do with the pregnenolone steal.
- PCOS: Progesterone is chronically low because high androgens — particularly testosterone — slow follicle development and prevent ovulation from occurring. The mechanism is at the level of the follicle, not the pregnenolone pathway.
- Chronic stress and HPA axis dysregulation: The brain deliberately reduces the hormonal signals driving ovulation when it perceives a state of extreme stress or energy deficit. This is a real mechanism — but it operates through GnRH suppression in the brain, not through pregnenolone competition in the adrenal gland.
Understanding which mechanism applies in your situation matters — because the clinical approach differs for each. The following sections cover each in detail.
Pregnenolone and Perimenopause: What's Really Happening
When patients in their 40s come to me with fatigue, sleep disruption, heavier periods, or mood changes, and they’ve been reading about the pregnenolone steal as a possible explanation, the actual picture is almost always rooted in the ovary rather than in any hormonal “theft.”
Here is what perimenopause actually looks like from a hormonal perspective. As women move through their late 30s and 40s, the ovaries gradually contain fewer follicles — and the follicles that remain are older, with different quality characteristics than those present in the reproductive years. This matters because ovulation, and specifically the corpus luteum that forms after ovulation, is the primary source of progesterone in a non-pregnant cycling woman. The corpus luteum is a temporary structure that forms from the follicle after the egg is released; it produces progesterone for approximately two weeks before it breaks down if pregnancy does not occur. No ovulation means no corpus luteum. No corpus luteum means no progesterone surge.
In perimenopause, two things change simultaneously. First, ovulation becomes less frequent — eggs don’t release as reliably as they did in the reproductive years, because the follicles are responding differently to FSH and LH stimulation. Fewer ovulations mean fewer corpus luteum cycles, which means progesterone production is reduced across the month. Second, even when ovulation does occur in perimenopause, the resulting corpus luteum often produces less progesterone than its counterpart in a younger ovary. The follicles in perimenopause do not behave the same way as follicles in the reproductive years — they are working in a different hormonal environment and with different egg quality.
The net result is a genuine decline in progesterone production — not because pregnenolone is being diverted, and not because of cortisol interference, but because the very structure responsible for progesterone production (the corpus luteum, formed from the follicle after ovulation) is forming less often and functioning differently when it does form. This is the natural biology of the ovarian transition through perimenopause.
How Declining Progesterone Affects Sleep
One of the more direct clinical consequences of this progesterone decline is disrupted sleep — and it is a pathway I discuss regularly with perimenopausal patients. Progesterone has a metabolite called allopregnanolone, which acts on GABA-A receptors in the brain — the same receptor system that benzodiazepines and many sleep medications act on. Allopregnanolone is a positive allosteric modulator of this receptor, meaning it enhances GABAergic inhibition and promotes feelings of calm, reduced anxiety, and the capacity to sleep.
When progesterone declines in perimenopause, allopregnanolone levels can decline with it. The resulting reduction in GABAergic tone is one of the clinical reasons perimenopausal women often notice worsening sleep quality, increased nighttime waking, heightened anxiety, and what can feel like PMS-like symptoms even outside of the luteal phase — sometimes in women who never experienced significant PMS during their reproductive years. For patients experiencing these symptoms, understanding the progesterone-allopregnanolone connection helps explain why the sleep disruption is hormonal in nature and not simply a response to life stress or anxiety. For a detailed look at how this mechanism plays out clinically, see this overview of how declining progesterone affects sleep in perimenopause at White Lotus Clinic.
How Declining Progesterone Affects Menstrual Bleeding
Reduced ovulation frequency in perimenopause also has a direct effect on menstrual bleeding patterns. Progesterone plays a key regulatory role in preparing the endometrium for a potential pregnancy — it transforms the endometrial lining from a proliferative state (driven by estrogen) into a secretory state (stable, differentiated, and ready for implantation). When progesterone is insufficient or absent due to anovulatory cycles, estrogen continues to stimulate endometrial growth without the counterbalancing secretory transformation that progesterone provides. This can result in heavier, longer, or more unpredictable periods — a pattern many perimenopausal patients describe as “my periods changed suddenly” or “my periods are much heavier than they used to be.” For a detailed clinical picture of this pattern, see heavy periods during perimenopause and the role of progesterone at White Lotus Clinic.
For women with PCOS, the progesterone picture carries an additional layer of complexity — because factors were already affecting follicle development and ovulation long before perimenopause began.
Pregnenolone and PCOS: A Different Mechanism
For women with PCOS who have been researching the pregnenolone steal as an explanation for their chronically low progesterone, the actual picture is different from what the steal theory describes — but it is no less important to understand.
In PCOS, the primary driver of low progesterone is not a pregnenolone pathway problem. It is an ovulation problem — and that ovulation problem is, in most cases, driven by elevated androgens.
How High Testosterone Affects Follicle Development
Many patients with PCOS have elevated testosterone levels. Testosterone and other androgens play a role in the early stages of follicle development, where low levels are actually beneficial — they help prime follicles to respond to FSH. But when androgen levels are chronically elevated, as they often are in PCOS, the developmental trajectory of the follicle changes. High testosterone slows follicle maturation and frequently arrests follicles before they reach the stage where they can ovulate. When ovulation doesn’t occur, the corpus luteum doesn’t form, and progesterone isn’t produced in any meaningful amount for that cycle.
This is not a pregnenolone steal. The pregnenolone pathway is not the site of disruption. The disruption is happening at the level of the follicle itself — in the way that elevated androgens alter the hormonal environment that follicles need to complete their development and release an egg.
Beyond the androgen effect on ovulation, the follicular environment in PCOS is also often characterised by elevated inflammatory markers. Inflammation within the follicle affects the granulosa cells that line it — these are the cells responsible for producing progesterone after ovulation. When the follicular environment is disrupted by chronically elevated androgens and inflammation, the granulosa cells’ ability to produce progesterone is affected even in cycles where some follicular development does occur.
PCOS With High Adrenal Androgens
In PCOS patients where the elevated androgens originate partly from the adrenal gland — often reflected in elevated DHEA-S on bloodwork — the mechanism is similar. Many of these patients also have elevated testosterone, and the effect on follicle development and ovulation follows the same pattern: testosterone-driven follicle arrest leading to reduced ovulation and, consequently, reduced progesterone. The source of the androgens (ovarian versus adrenal) matters for the clinical approach to managing them, but the downstream effect on ovulation and progesterone production is comparable.
PCOS Entering Perimenopause
The clinical picture becomes more complex for PCOS patients approaching perimenopause. In non-PCOS patients, perimenopause introduces a gradual ovarian aging process — reduced follicle quality and quantity leading to less frequent ovulation and declining progesterone. In PCOS patients, that natural perimenopause-related reduction in ovulation is added on top of an already-reduced ovulation frequency driven by androgen excess. The result is that PCOS patients entering perimenopause often experience a compounding of two mechanisms that both reduce progesterone — and the clinical assessment needs to account for both.
If you have PCOS and are noticing changes in your cycle, energy, or hormone patterns as you move through your late 30s or 40s, the intersection of these two mechanisms is worth understanding in detail. PCOS and perimenopause — the compounded hormonal picture at White Lotus Clinic covers the clinical implications of this overlap.
What About Cortisol? The Real Relationship Between Stress and Your Hormones
Even if the pregnenolone steal is not the mechanism, stress and cortisol do have real effects on reproductive hormones — and it is worth addressing this directly, because many patients who encounter the pregnenolone steal concept are also experiencing symptoms they associate with stress, adrenal fatigue, or HPA axis dysregulation.
Cortisol can, in fact, decrease progesterone, but not by stealing pregnenolone. The mechanism is through the brain. Cortisol, along with corticotropin-releasing hormone (CRH) and other stress hormones, suppresses GnRH pulsatility in the hypothalamus. Reduced GnRH pulsatility means reduced LH and FSH secretion from the pituitary, which in turn means reduced follicle stimulation, reduced ovulation, and reduced progesterone production from the corpus luteum. The entire suppression pathway is neurological and hormonal — it originates in the brain and acts through the pituitary, not through a peripheral competition for pregnenolone substrate.
The most important thing to understand about this relationship is that it is deliberate. As I explain to patients: reproduction is one of the most metabolically demanding functions the body undertakes. The human brain has evolved to make a calculated energy trade-off during periods of extreme stress or food scarcity — temporarily reducing the hormonal investment in reproduction in order to direct resources toward survival. This is not your hormones stealing from each other. It is your brain making a purposeful decision, prioritising immediate survival over reproductive function. When the stressor resolves, and when energy availability returns to normal, the reproductive axis can recover.
When Cortisol Assessment Is Clinically Relevant
Assessing cortisol is a separate question from the pregnenolone steal — the two are not directly linked. In clinical practice, the pattern that prompts me to assess cortisol is when multiple hormones are simultaneously low. If bloodwork shows low LH, low FSH, low estradiol, and low progesterone — an all-low pattern across both pituitary and sex hormones — that can suggest the brain is in an energy-conservation state and reducing the hormonal investment in reproduction broadly. In that context, assessing cortisol helps build a more complete picture of how the HPA axis is functioning.
Cortisol can be assessed through several methods: a 24-hour urine cortisol collection, a blood cortisol drawn in the morning (when cortisol should be at its daily peak), or a salivary cortisol panel taken at multiple points across the day to capture the diurnal rhythm. The choice between these depends on the clinical picture and what information is most useful to guide next steps.
For a detailed overview of how HPA axis function and cortisol patterns are assessed in the context of hormonal symptoms, see HPA axis assessment and adrenal function at White Lotus Clinic.
Should You Take Pregnenolone Supplements?
Because the pregnenolone steal theory frames pregnenolone as a depleted resource, the natural extension of the theory is that supplementing with pregnenolone will replenish the supply and correct downstream hormonal deficits. Pregnenolone supplements are widely available over the counter, particularly in the United States, and some patients arrive at consultations already taking them.
My clinical perspective on this is straightforward: hormones do not work like a linear production line where adding more raw material predictably increases downstream output. They operate on feedback loops. The body continuously monitors circulating hormone levels and adjusts production, sensitivity, and clearance in response. Adding pregnenolone as a substrate does not reliably translate into increased production of a specific downstream hormone like progesterone, DHEA, or estrogen — because whether and how that conversion occurs depends on enzyme expression, receptor availability, tissue-specific regulatory signals, and feedback dynamics that a substrate supplement cannot control.
In practice, I rarely see patients who are taking pregnenolone — it is less common than the volume of online discussion might suggest. When it does come up, my approach is to redirect the conversation toward a more clinically useful question: which specific hormone appears to be deficient, and why? Understanding what is actually driving the deficiency — whether it is perimenopause-related anovulation, PCOS-related androgen excess, HPA axis-driven suppression, or something else — points toward a specific intervention rather than a non-specific substrate supplement.
For patients whose progesterone is confirmed to be low, the clinical options are either to support the conditions that make ovulation more likely (relevant in PCOS, where addressing androgen excess and insulin resistance can restore ovulatory frequency) or to replace progesterone directly. Ontario naturopathic doctors have prescribing authority for bioidentical hormones including progesterone — which means that when a comprehensive hormonal assessment indicates progesterone deficiency, there is a specific, evidence-informed treatment pathway available. For more detail on this approach, see cyclic progesterone therapy for progesterone deficiency at White Lotus Clinic.
Frequently Asked Questions
Is the pregnenolone steal real?
The simplified version of the pregnenolone steal — the idea that stress diverts a shared pool of pregnenolone toward cortisol and away from sex hormones — is not supported by how steroidogenesis actually works. Each steroidogenic tissue (the adrenal glands, the ovaries) produces its own pregnenolone independently from cholesterol, under its own regulatory signals. There is no shared reservoir to divert. However, the underlying clinical observation that the pregnenolone steal attempts to explain is real: stress, perimenopause, and PCOS do reduce progesterone and other sex hormones. The mechanism is more specific and more complex than the simplified steal narrative suggests, but the symptoms patients are trying to explain are genuine.
Why is my progesterone low in perimenopause?
In perimenopause, progesterone declines primarily because ovulation becomes less frequent. The main source of progesterone in a cycling woman is the corpus luteum — a temporary structure that forms from the follicle after ovulation and produces progesterone for approximately two weeks. As egg quality and ovarian reserve change during perimenopause, follicles respond differently to FSH and LH, ovulation occurs less often, and the corpus luteum forms less frequently. Even in cycles where ovulation does occur, perimenopausal follicles often produce a corpus luteum that secretes less progesterone than would be expected in younger ovaries. This is the biology of the ovarian transition — it is not a pregnenolone steal.
Does stress actually lower progesterone?
Yes — but through the brain, not through pregnenolone competition. Chronic or significant stress activates the hypothalamic-pituitary-adrenal (HPA) axis, and the stress hormones involved — particularly corticotropin-releasing hormone (CRH) — directly suppress the pulsatile release of GnRH from the hypothalamus. Reduced GnRH pulsatility leads to reduced LH and FSH from the pituitary, which reduces follicle stimulation, reduces ovulation frequency, and reduces progesterone production. This is a deliberate, centrally mediated response. It is also worth noting that the progesterone produced by a normal ovulatory cycle is substantially greater in volume than any pregnenolone diversion could account for — which is another reason the steal mechanism does not hold up as the primary explanation.
Does PCOS affect the pregnenolone pathway?
In PCOS, the main reason progesterone is low is not a pregnenolone pathway disruption — it is an ovulation problem driven by elevated androgens, particularly testosterone. High testosterone slows follicle maturation and often stops follicles before they can ovulate, which means the corpus luteum never forms and progesterone is not produced. Inflammation in the follicular environment — common in PCOS — further affects the ability of granulosa cells to produce progesterone. For patients with PCOS who also have elevated adrenal androgens (reflected in elevated DHEA-S), many of these patients also have elevated testosterone, and the mechanism is similar: elevated androgens affecting follicle development and ovulation frequency, with downstream effects on progesterone.
Should I take pregnenolone supplements?
Pregnenolone supplements are widely available, but hormones operate on feedback loops rather than linear production pathways. Adding pregnenolone as a substrate does not reliably increase a specific downstream hormone like progesterone, because the conversion depends on enzyme activity, regulatory signals, and feedback dynamics that a substrate supplement cannot direct. A more clinically useful approach is to identify which specific hormone is deficient and why — and then either support the conditions that make ovulation more likely, or consider replacing the specific hormone that is low. If you are already taking pregnenolone, that is worth discussing in a comprehensive hormonal assessment where your full picture can be evaluated.
What tests assess the pregnenolone pathway?
In clinical practice, assessing the downstream hormones — progesterone, estradiol, LH, FSH, DHEA-S, and testosterone — tends to be more clinically actionable than measuring pregnenolone directly. These downstream values reveal where in the hormonal cascade a problem is occurring and what is driving it. For patients where stress and HPA axis function are relevant, cortisol can be assessed in several ways: a 24-hour urine cortisol collection, a morning blood cortisol (taken when cortisol should be at its daily peak), or a salivary cortisol panel taken at multiple points across the day to capture the diurnal rhythm. Pregnenolone levels are not typically tested as part of a standard hormonal assessment.
What is the difference between perimenopause and stress-related low progesterone?
Both perimenopause and chronic stress can result in low progesterone and irregular cycles, but the mechanisms and lab patterns tend to differ. In perimenopause, FSH typically rises over time as the pituitary works harder to stimulate aging follicles, while estradiol can fluctuate widely before eventually declining. In stress-related suppression of the reproductive axis, the pattern is often one where LH, FSH, estradiol, and progesterone are all low together — a broad hormonal suppression rather than the rising FSH and fluctuating estrogen that characterises perimenopause. A hormonal assessment can help clarify which picture is relevant in a given situation, which in turn informs the clinical approach.
About Dr. Fiona McCulloch, ND
Dr. Fiona McCulloch is a naturopathic doctor with a Fellowship in the American Board of Naturopathic Endocrinology — a fellowship focused on the assessment and management of endocrine and hormonal conditions, including the steroidogenesis pathways discussed in this article. She is the author of 8 Steps to Reverse Your PCOS and served as a peer reviewer for the 2023 International Evidence-based PCOS Guidelines. She practises at White Lotus Clinic in Toronto, where she has worked in hormonal health for over 25 years.
As naturopathic doctors in Ontario, Dr. Fiona and her colleagues hold prescribing authority for bioidentical hormones including progesterone — meaning that a comprehensive hormonal assessment has a direct treatment pathway when progesterone deficiency is identified and treatment is indicated.
- Doctor of Naturopathic Medicine (ND)
- Fellow, American Board of Naturopathic Endocrinology
- Author: 8 Steps to Reverse Your PCOS
- Peer reviewer: 2023 International Evidence-based PCOS Guidelines
- 25+ years in hormonal health
- White Lotus Clinic, Toronto — Ontario ND prescribing authority


When a Hormonal Assessment Makes Sense
If the mechanisms discussed in this article resonate with what you are experiencing — irregular or changing cycles, declining progesterone, sleep disruption, heavier periods in perimenopause, or long-standing low progesterone in PCOS — a comprehensive hormonal assessment can help clarify the picture. At White Lotus Clinic, that assessment looks at downstream hormones and pituitary signals (LH, FSH, estradiol, progesterone, DHEA-S, cortisol) to identify where in the hormonal cascade a disruption is occurring and what approaches may be relevant in your specific situation.
If you are in perimenopause or approaching it, and your cycles, sleep, or mood have been changing:
If you have PCOS and are noticing shifts in your cycle, energy, or hormonal patterns — especially as you move into your late 30s or 40s:
References
- Miller WL, Auchus RJ. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocrine Reviews. 2011;32(1):81–151. [Compartmentalization of steroidogenesis; StAR protein as rate-limiting step]
- Kalantaridou SN, Makrigiannakis A, Zoumakis E, Chrousos GP. Stress and the female reproductive system. Journal of Reproductive Immunology. 2004;62(1–2):61–68. [CRH-mediated GnRH suppression; hypothalamic amenorrhea mechanism]
- Bäckström T, Andersson A, Andréen L, et al. Role of the GABAergic system in the pathophysiology of premenstrual syndrome. Neuropsychopharmacology. 2003;28(Suppl 1):S29–S34. [Allopregnanolone / GABA-A receptor pathway; progesterone → sleep mechanism] [NEEDS SOURCE: more current allopregnanolone-sleep citation preferred]
- Santoro N, Randolph JF Jr. Reproductive hormones and the menopause transition. Obstetrics and Gynecology Clinics of North America. 2011;38(3):455–466. [Perimenopause ovarian aging; progesterone decline pattern]
- Dumesic DA, Oberfield SE, Stener-Victorin E, Marshall JC, Laven JS, Legro RS. Scientific statement on the diagnostic criteria, epidemiology, pathophysiology, and molecular genetics of PCOS. Endocrine Reviews. 2015;36(5):487–525. [PCOS androgen excess; folliculogenesis; theca cell hyperactivity]
- McCulloch F. Clinical perspective on pregnenolone pathway assessment and progesterone deficiency management. Recorded interview, March 28, 2026. [All clinical observations regarding perimenopause mechanism, PCOS mechanism, cortisol assessment, and supplement approach]